 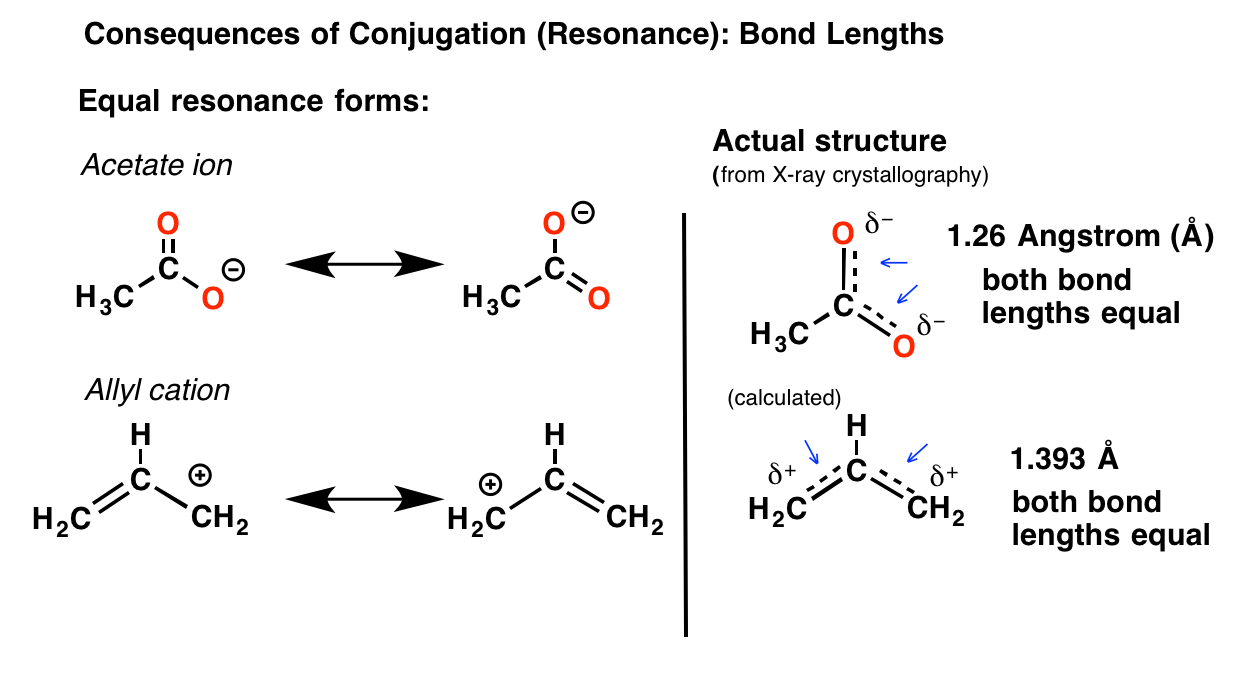
The positive resonance effect examples are -OH, -OR,-SH, and -SR. Usually, the groups are denoted by +R or +M - the molecular electron density increases in this process. The positive resonance effect happens when the groups release electrons to the other molecules by the delocalization process. Two types of Resonance effects exist, namely: 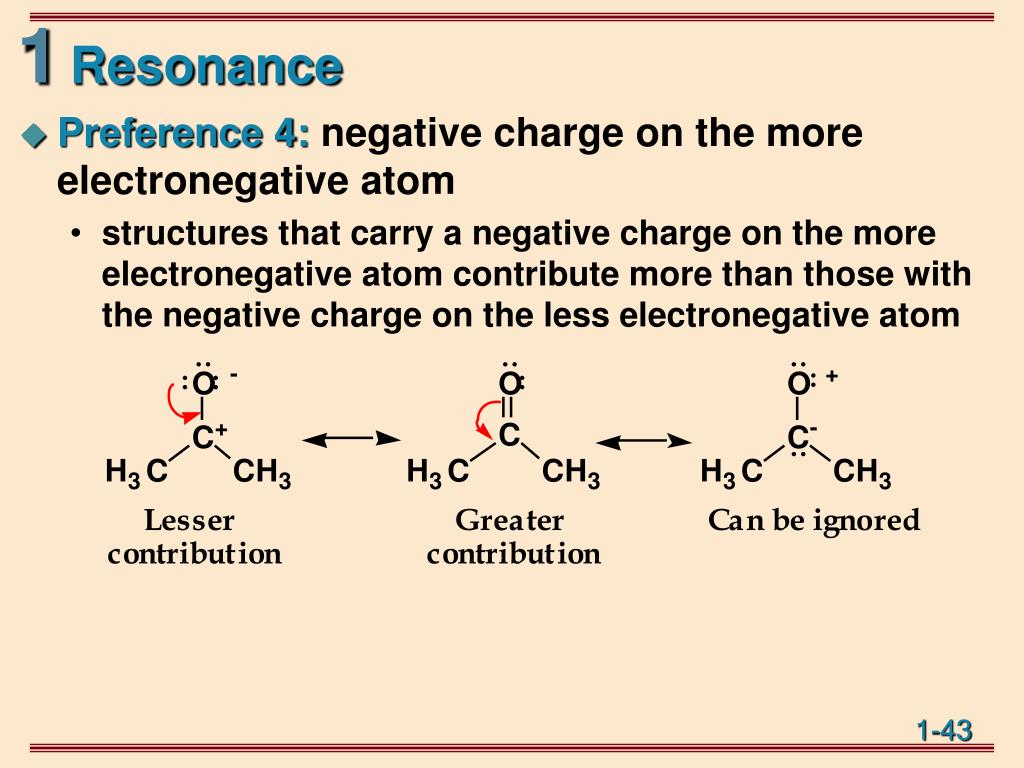
The organic compounds have these double bonds in the structures and have the overlapping of the p-orbitals, usually on the two adjacent sides of carbon atoms. The resonance effect definition can be given as a chemical phenomenon, observed in the characteristic compounds containing double bonds in the organic compounds. Resonance in Chemistry helps to understand the stability of a compound along with the energy states.ĭefinition Of Resonance Effect In Chemistry In sim ple words, reson ance is the molecule with multiple Lewis structures. It also occurs by the interaction of 2 pi bonds present in the adjacent atoms. The definition of the resonance effect explains the polarity caused by the interaction between the electron lone pair and the pi bond in a molecule. The above representation shows various resonance structures of different compounds with the respective resonance effects. The symbols M or R are used to represent the resonance effect. The withdrawal or releasing effect of electrons attributed to a specific substituent through the delocalization of π or pi-electrons, which can be seen by drawing different canonical structures, is known as a resonance or mesomeric effect. Resonance Effect Or Mesomeric Effect In Chemistry A molecule or ion with these delocalized electrons can be described by contributing various structures known as resonance structures. The factor called Mesomerism or resonance in Organic Chemistry explains the delocalized electrons within certain molecules where a single Lewis structure does not express the bonds. The organic molecules also exhibit resonance or mesomerism properties. However, they do not prevent the organic compounds from taking on the various properties of their physical characteristics and chemical reactivity. Many biological molecules consist of a combination of these six elements: nitrogen, carbon, hydrogen, sulphur, oxygen, and phosphorus. Diversely, all these factors relate to the organic molecules. The electronic factors influencing the organic reactions include the inductive effect, the electromeric effect, resonance effects, hyperconjugation, and more. The resonance effect in Organic Chemistry is the electron behaviour differs when the elements other than the hydrogen and carbon atoms take part in the formation of molecular bonds actively. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
